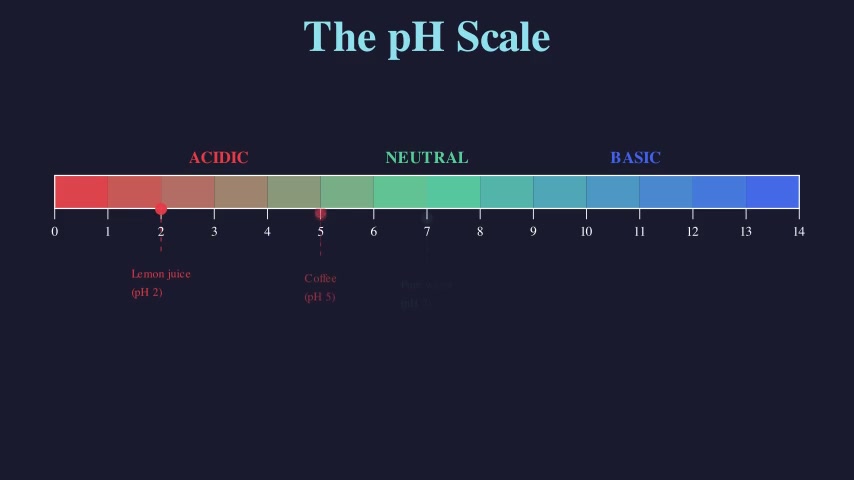
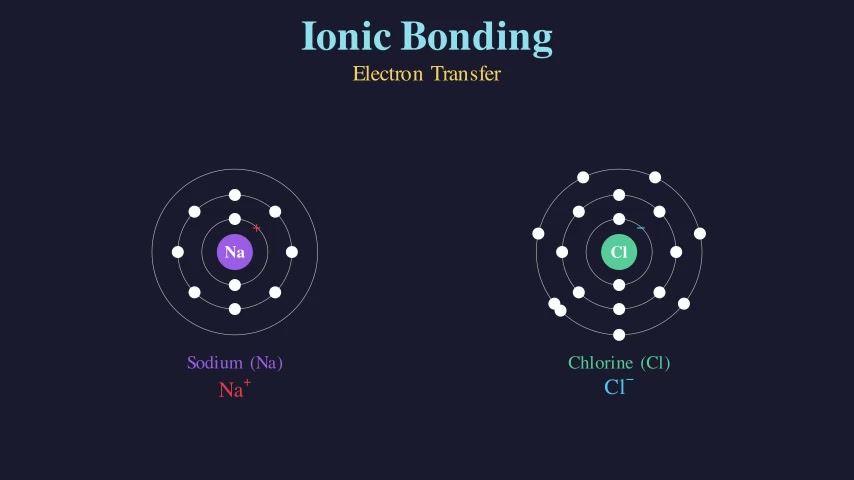
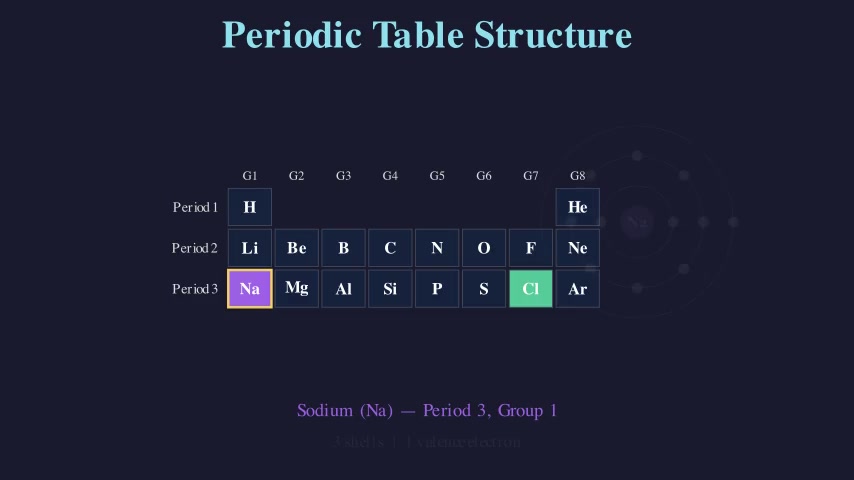
Balancing Chemical Equations
Teaches the law of conservation of mass by balancing two chemical equations step by step. Shows atom-count tracking on both sides of the equation, then demonstrates adding coefficients to balance H₂ + O₂ → H₂O and CH₄ + O₂ → CO₂ + H₂O.